
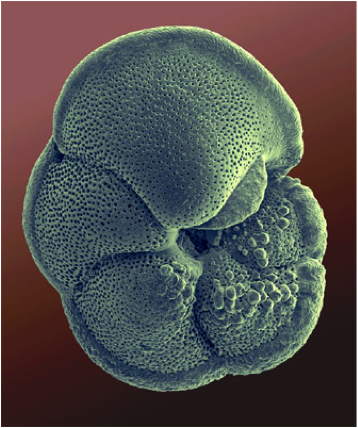
Foraminifera (Globigerinoides ruber, Globorotalia menardi) (photos ©Dee Breger, Micrographic Arts)
The sediments that accumulate on the ocean floor are of many types with various compositions. Among the sediment particles created by plankton living in the upper sun-lit waters are small shells made of calcium carbonate (CaCO3) that we call calcite. These shells (typically as large as a grain of beach sand) are secreted by both microscopic animals (foraminifera) and plants (coccolithophores or coccolithophorids).


Foraminifera (Globigerinoides ruber, Globorotalia menardi) (photos ©Dee Breger, Micrographic Arts)
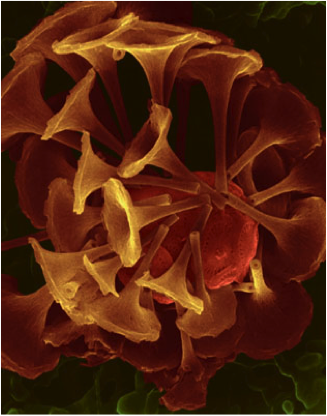

Coccolithophores (right: Emiliania huxleyi) (photos ©Dee Breger, Micrographic Arts)
Foraminifera (foraminifers or, informally, just forams) are single-celled amoeboid protists. Forams are abundant all across the global ocean. They either live on the sea bottom (benthic) or float in the upper water column (planktonic). Of the estimated 4000 species living today, 40 are planktonic. Coccolithophores are very tiny and are commonly called nannoplankton. Coccolithophores are almost exclusively marine and occur in abundances throughout the surface sun-lit portion of the ocean called the euphotic zone.
The shells of foraminifera and coccolithophores accumulate as calcium carbonate rich sediment called calcaeous ooze and calcareous marl. The calcareous oozes and marls comprise almost 25% of all deep-sea sediments by volume. The global distribution and abundance of calcareous sediment is highly variable and reflects primarily variable preservation of the shell material from dissolution by seawater. The calcite shell material is more soluable in cold water and at high pressure. Thus the degree of dissolution and loss of shell material increases with depth in the ocean. Marine organisms produce calcite at roughly 4 times the rate at which the ingredients for this mineral are supplied to the ocean by continental weathering and planetary outgassing from volcanic activity.
Since aurface waters are supersaturated with respect to calcite (CaCO3), shells are well-preserved in shallow regions of the continenal shelves and oceanic plateaus. However, deep waters are undersaturated. As a result a transition zone (called the calcium compensation depth or CCD) separates the ocean floor above 4000 meters depth where calcite is largely preseved from the abyssal ocean at 5000 to 6000 meters where calcite is almost entirely dissolved. The map of the precent of calcium carbonate distribution in the ocean floor sediment carpet reveals higher percentages of calcite on the shallower elevations of the mid-ocean ridges and much lower percentages on the basin floors and abyssal plains.
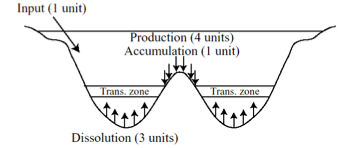 From Broecker, 2004.
From Broecker, 2004.
The ultimate burial of carbonate sediments is dependent upon the supply of calcium to the ocean by freshwater that leaches calcium from continental rocks during chemical weathering. The residence time of calcium in the ocean is the ratio of the oceanic calcium inventory to the supply of calcium via rivers. The residence time is about 1 million years. Therefore, the net rate of calcite burial does not change over million year timescales, but the burial rate has changed appreciably from basin to basin and region to region as climate has varied from warm (interglacial) to cold (glacial) and back to warm.
The CO2 (dissolved gas) in sea water effects the alkalinity of the ocean. As the ocean absorbs more CO2 from the atmosphere, the pH of seawater decreases and the ocean becomes more acidic. A more acidic ocean is less solubable and carbonate sediments will be only be preserved at somewhat shallower depths. Thus the global calcium carbonate distribution as showed in this map will change with the continued addition of anthropogenic CO2 to the atmosphere and ocean.
Data Citations:
Published articles of interest: